Potential Medicines- Testing Plant Substances
May 15, 2015- May 16, 2015
Purpose: The purpose of this lab is to find active plant ingredients found locally that can inhibit bacterial growth.
Materials: LB broth, Media bottles, Sterilizer, Hot water bath, LB agar, Laminar flow hood, Disinfectant, Safety glasses, Bunsen burner, Inoculating loop(Ni/Cr wire), Petri dishes, Stock plate, Plant sample, Mortar and pestle, Pipet(10 mL, 1 mL), Short-stemmed plastic funnels), Filter paper discs(5 mm), Beakers(100 mL), Syringe and filter, Reaction tubes, Tube racks, Methanol, Heat block, Dry block heater, Ampicillin, Glass spreader, Incubator oven, Tweezers, Alcohol
Procedure Day One: First, the mortar and pestle is used to grind two grams of plant tissue and combine it with ten milliliters of deionized water. The solution rests for three minutes before filtering the sample through a filter paper funnel. Then, a syringe filter is used to extract the filtered sample to sterilize it. A one milliliter sample of the extract is transferred to a microtube and labelled. These steps are repeat with methanol in place of water to extract different components. Let the extract sit on heat blocks for twenty-four hours to evaporate the methanol.
Procedure Day Two: Reconstitute the sample with methanol by adding one milliliter of sterilized water. For both samples of extract, place two filter paper discs into the test tubes. In order to ensure the filter papers are sterile, use flames to burn off an alcohol soaked pair of tweezers. To set up controlled results, place 0.5 milliliters of sterilized water to one test tube for the negative control and 0.5 milliliters of ampicillin for the positive result. In each test tube, add one sterilized filter paper disc using the same methods of flame sterilization. These samples are then place in a refridgerator over night in order to soak up the extract.
Procedure Day Three: Label a petri dish to form four quadrants: two water and two methanol sections. In the center of dish, label for positive and negative controls(one of each). After labeling,
place one milliliter of E-coli bacteria onto a petri dish that is covered in Agar. Using a flame sterilized spreader, cool the bar on the side of the Agar before spreading the E-coli around to completely cover the dish. Let the bacteria rest and soak into the Agar for fifteen minutes. Then, use flame sterilized tweezers to place one soaked filter paper disc on each designated section. If needed, on the lid of the petri dish, use two sterile tweezers to separate the discs so only one is placed into each quadrant. After covering the petri dish with the cover, incubate at thirty-seven degrees Celsius for twenty-four hours.
place one milliliter of E-coli bacteria onto a petri dish that is covered in Agar. Using a flame sterilized spreader, cool the bar on the side of the Agar before spreading the E-coli around to completely cover the dish. Let the bacteria rest and soak into the Agar for fifteen minutes. Then, use flame sterilized tweezers to place one soaked filter paper disc on each designated section. If needed, on the lid of the petri dish, use two sterile tweezers to separate the discs so only one is placed into each quadrant. After covering the petri dish with the cover, incubate at thirty-seven degrees Celsius for twenty-four hours.
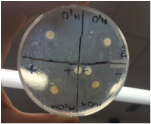
Results and Analysis: Unfortunately, none of the filter papers showed any positive results, not even the positive control. There was about a tenth of a centimeter between the controlled results and the start of bacterial growth. The water discs showed the same results of providing a very thin ring that was a tenth of a centimeter wide. The methanol discs showed slightly larger rings, but the size was still not wide enough to show positive anti-bacterial results. The rings stretched to reach approximately three tenths of a centimeter before the bacteria continued to grow. I expected the positive control to at least show a wider ring, but the data was the same as the negative control or even slightly smaller.
There are several possible errors that could have effected the results. For example, the liquid on the soaked filter papers could have pushed the bacteria backwards a little while being placed onto the petri dish. This resulted in a slight bubble around the ring, but next to the filter paper, the bacteria still grew. Another potential issue that could have effected the lab was a mistake in the sterilization processes. There were many steps required in the procedure that proper sterilization techniques were essential; this leaves room for many opportunities of failure and accidentally bringing unwarranted bacteria or alcohol into the results.
There are also improvements that can be made to this experiment or an extension of this lab to further test the anti-bacterial plants in hopes of finding an effective medicine. Personally, my results showed all negative results so I would redo the exact lab making sure to clearly label and process each sample or extract. I would probably spend more time making sure the plant extracts were fully crushed and filtered through the paper. Another change I would make to the current experiment would be to spread out the filter paper discs more to ensure different discs don't interfere with each other. If I obtained positive results, it would be ideal to retest the plant material with at least five filter paper discs to have statistical proof that the extract is anti-bacterial. After this, a potential use of a variety of alcohol to isolate multiple types of anti-bacterial material from the same plant. These are only some of the possible future experiments before actual production and more complete isolation of the plant material is performed. Because he hasn't botany
There are several possible errors that could have effected the results. For example, the liquid on the soaked filter papers could have pushed the bacteria backwards a little while being placed onto the petri dish. This resulted in a slight bubble around the ring, but next to the filter paper, the bacteria still grew. Another potential issue that could have effected the lab was a mistake in the sterilization processes. There were many steps required in the procedure that proper sterilization techniques were essential; this leaves room for many opportunities of failure and accidentally bringing unwarranted bacteria or alcohol into the results.
There are also improvements that can be made to this experiment or an extension of this lab to further test the anti-bacterial plants in hopes of finding an effective medicine. Personally, my results showed all negative results so I would redo the exact lab making sure to clearly label and process each sample or extract. I would probably spend more time making sure the plant extracts were fully crushed and filtered through the paper. Another change I would make to the current experiment would be to spread out the filter paper discs more to ensure different discs don't interfere with each other. If I obtained positive results, it would be ideal to retest the plant material with at least five filter paper discs to have statistical proof that the extract is anti-bacterial. After this, a potential use of a variety of alcohol to isolate multiple types of anti-bacterial material from the same plant. These are only some of the possible future experiments before actual production and more complete isolation of the plant material is performed. Because he hasn't botany
Thinking Like a Bio-technician:
1. If an extract shows negative for antimicrobial assay, it does not necessarily mean that the extract is a antimicrobial agent. For example, the molecules that are polar could have antimicrobial attributes, but these molecules would not be present if the plant material was combined with alcohol. Due to polar molecules matching polar and vice-versa, certain extracts will show negative results even if the plant is an antimicrobial agent. There are also human errors that could have occurred and shown inaccurate results for negativity.
2. Some of the samples with methanol extractions could possible smell like alcohol. This is an issue because alcohol kills bacteria. This addition could interfere with the control of the experiment because then the results won't show the reason for lack of bacterial growth. You wouldn't be able to record whether the death of bacteria was caused by the substance in the extract or the alcohol.
3. In extracts with more than one compound, there are certain steps needed to isolate each compound in order to test each individually to figure out which is causing the antimicrobial activity. One way to do this is to use chromatography to separate the layers depending on the compounds polarity. This type of chromatography is called thin layer chromatography because it layers the compounds based on which they are polar, like water, or non-polar, like alcohol. This would separate each compound as it runs through the columns and then you could test each one for antimicrobial activity individually.
1. If an extract shows negative for antimicrobial assay, it does not necessarily mean that the extract is a antimicrobial agent. For example, the molecules that are polar could have antimicrobial attributes, but these molecules would not be present if the plant material was combined with alcohol. Due to polar molecules matching polar and vice-versa, certain extracts will show negative results even if the plant is an antimicrobial agent. There are also human errors that could have occurred and shown inaccurate results for negativity.
2. Some of the samples with methanol extractions could possible smell like alcohol. This is an issue because alcohol kills bacteria. This addition could interfere with the control of the experiment because then the results won't show the reason for lack of bacterial growth. You wouldn't be able to record whether the death of bacteria was caused by the substance in the extract or the alcohol.
3. In extracts with more than one compound, there are certain steps needed to isolate each compound in order to test each individually to figure out which is causing the antimicrobial activity. One way to do this is to use chromatography to separate the layers depending on the compounds polarity. This type of chromatography is called thin layer chromatography because it layers the compounds based on which they are polar, like water, or non-polar, like alcohol. This would separate each compound as it runs through the columns and then you could test each one for antimicrobial activity individually.
