|
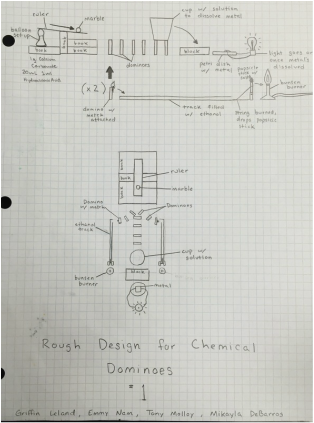
The goal of this project was to connect a series of chemical reactions in order to create a Rube Goldberg machine; the end goal being lighting an LED. There were certain requirements such as using a lever, destroying a metal, and adding at least other type of reaction. Our group decided to start with creating a gas to rise into a balloon and tip a lever. Rest on a notch on the lever, a marble would then fall into a chute before knocking over a domino. This domino would create a chain reaction of falling dominoes that would branch off in two different directions. One the first track the end result would be lighting salts over a bunsen burner to produce a vibrant flame. On the other end a solution would break a metal in order to cause a circuit to light up the LED. Although the physics aspects were more challenging to get precise and connect
|
together, there were chemistry topics that were studied. We incorporated the chemistry ideas used in the labs and previous knowledge to create our final product.
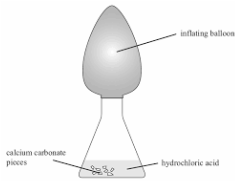
Preceding this project, there was a series of labs that covered several topics and introduced potential ideas for chemical reactions that could be utilized. First of all, I learned how to distinguish different liquids depending on their reactions with other elements. For example, copper chloride was capable of dissolving aluminum foil, but hydrochloric acid was unable to do so. With an unknown solution, if it was placed on aluminum and destroyed it, then I could deduce that it was the copper chloride. Another topic we covered was the different types of chemical reactions: synthesis, decomposition, combustion, single replacement, and double replacement. In order to learn this, I combined specific liquids and noted the products. By writing the balanced chemical equations and comparing the reactants to the products, I was able to visualize each type of reaction. Through this lab I was also able to discern the necessary parts needed to make each type of reaction possible. Finally, I also studied various methods to produce carbon dioxide and compared the techniques. Placing calcium carbonate and hydrochloric acid into an erlenmeyer flask then quickly sealing a balloon onto the top gave me the ability to measure the amount of gas produced over a certain amount of time. Vinegar and baking soda was another combination that formed gas. Also, if calcium chloride alone was added to the flask, then heated with a hot plate, a gas was formed and expanded.
Step One:Calcium Chloride and Hydrochloric Acid Reaction2 HCl +Ca(OH)3 ---> CaCl2 + CO2 + H2O
The first step was to mix 1 gram of calcium carbonate with 10 milliliters of hydrochloric acid. This formed carbon dioxide in gas form and two other products of water and In order to ensure all the carbon dioxide was captured, we placed the calcium carbonate and hydrochloric acid into the balloon and erlenmeyer flask respectively. Then, once the trial was prepared to run, the balloon could just be tipped over, resulting in the pouring of calcium carbonate into the container. This set up was placed directly under one side of the lever and caused the rising balloon to set off the first steps of the chemical dominoes into action.
|
Step Two:Ethanol Combustion ReactionC2H6O + 3(O2) ---> 3(H2O) + 3(CO2)
The tipping lever eventually lead to the next section of the string of reactions: a combustion reaction utilizing ethanol and oxygen. Combustion reactions occur when a reactant of a hydrocrabon reacts with the oxygen in the atmosphere and forms products of water vapor and carbon dioxide. In order to fit with the chemical dominoes, we taped a lit match to the end of a domino prior to setting off the initial reaction. The slow burning match was still lit as the lever caused a marble to knock down the preceding dominoes. The match fell over, connecting with a string that was soaked in alcohol. By setting the string on a track, the direction of the flame traveled in a straight path, directly towards the next reaction.
|
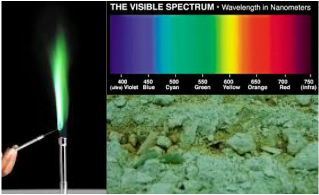
Step Three:Burning Copper(II) Chloride to Produce Colored FlameCuCl2 + Energy --> Emittance of Photons
The burning string was directly attached to a length of ribbon. This fragile ribbon was tied to the end of a popsicle stick that was suspended at an acute angle over a bunsen burner. This resulted in the stick falling due to gravity directly onto the bunsen burner once the tension from the ribbon disappeared. The edge of the popsicle stick would collide with the base of the flame, keeping the tip of the stick burning in the fire. In order to create a chemical reaction, we secured powdery copper(II) chloride salt onto the end of the popsicle stick. When passed through a flame, the electrons in the copper(II) chloride become "excited" by the heat and jumped to a higher energy level. However, the electron would soon fall back down to the resting state. This process causes a photon to be released from the loss of excess energy. Depending on the distance the electrons fall, the wavelength of the photon will change, directly corresponding with the color of flame that is produced.
|
Step Four:2 Al + 3 Cu(Cl)2 ---> 3 Cu + 2 Al(Cl)3
Breakdown of AluminumThe circuit lit up the LED because the aqueous solution of copper chloride dissolved the aluminum. It forms copper in solid form and aluminum chloride as an aqueous solution. Since the aluminum is degraded, the portion of the circuit is broken. This allows the resistance to decrease and enough voltage to flow and power the LED.
|
*The circuit worked, but the battery connector was not secured, so that is why the LED lit up after holding the battery.
Reflection: This project was relatively long so there were many opportunities to learn from the experience. One portion of the project that I struggled with was connecting all the steps properly in the same trial run. One way to alleviate this challenge would be to thoroughly test each section of the steps and to set up the connections to ensure the chemical reaction The physics section of the construction of the series was more challenging than measuring specific parts of chemicals. Combining reactants, making more concentrated versions of solutions, and dissolving materials were relatively simple because we were able to experiment with these factors prior to adding them into our system. Therefore, I believe it would be beneficial to address the physical construction of the machine and spend more time focusing on that aspect. This challenge taught me to be concerned with all aspects of a project, even if that attribute seems flawless in hypothetical circumstances. Another aspect that was a struggle was needing to reset the entire system each project work day. It took a chunk of time out of our day because we would need to completely need to replace each element. I learned that using materials that can be renewable or recycled is easier and more efficient. When possible, it would be ideal to use resources that are the most accessible.
The project also succeeded in several areas. First of all, since our system could be split into a couple of different steps, my group was able to work on each aspect of the machine separately. This means we could spend more time refining very specific parts of the design or perfect a step. We wouldn’t need to worry about ruining another part of the set-up because the connections could be split. Another part of the project that was a positive aspect was the creativity. Typically, it is challenging to differentiate from other groups, but this project gave our group a chance to connect each reaction in a unique method. It was exciting to see which ideas were too unrealistic, while others that I was skeptical about seemed to work out well. This taught me to be more open-minded and experiment with something before automatically shooting it down.
The project also succeeded in several areas. First of all, since our system could be split into a couple of different steps, my group was able to work on each aspect of the machine separately. This means we could spend more time refining very specific parts of the design or perfect a step. We wouldn’t need to worry about ruining another part of the set-up because the connections could be split. Another part of the project that was a positive aspect was the creativity. Typically, it is challenging to differentiate from other groups, but this project gave our group a chance to connect each reaction in a unique method. It was exciting to see which ideas were too unrealistic, while others that I was skeptical about seemed to work out well. This taught me to be more open-minded and experiment with something before automatically shooting it down.